Clinical Laboratory Services Market to Hit USD 352 Bn, Globally, by 2032 at 5.3% CAGR | Data By Market.us
Page Contents
Market Overview
Published Via 11Press : The clinical laboratory services market encompasses various diagnostic tests and services used for disease diagnosis. This market includes services in areas like hematology, microbiology, immunology, molecular diagnostics, and clinical chemistry among others. As chronic illnesses like cancer and diabetes become more commonplace, demand for diagnostic testing services has also grown due to rising healthcare expenditure and technological developments that support them both directly and indirectly.
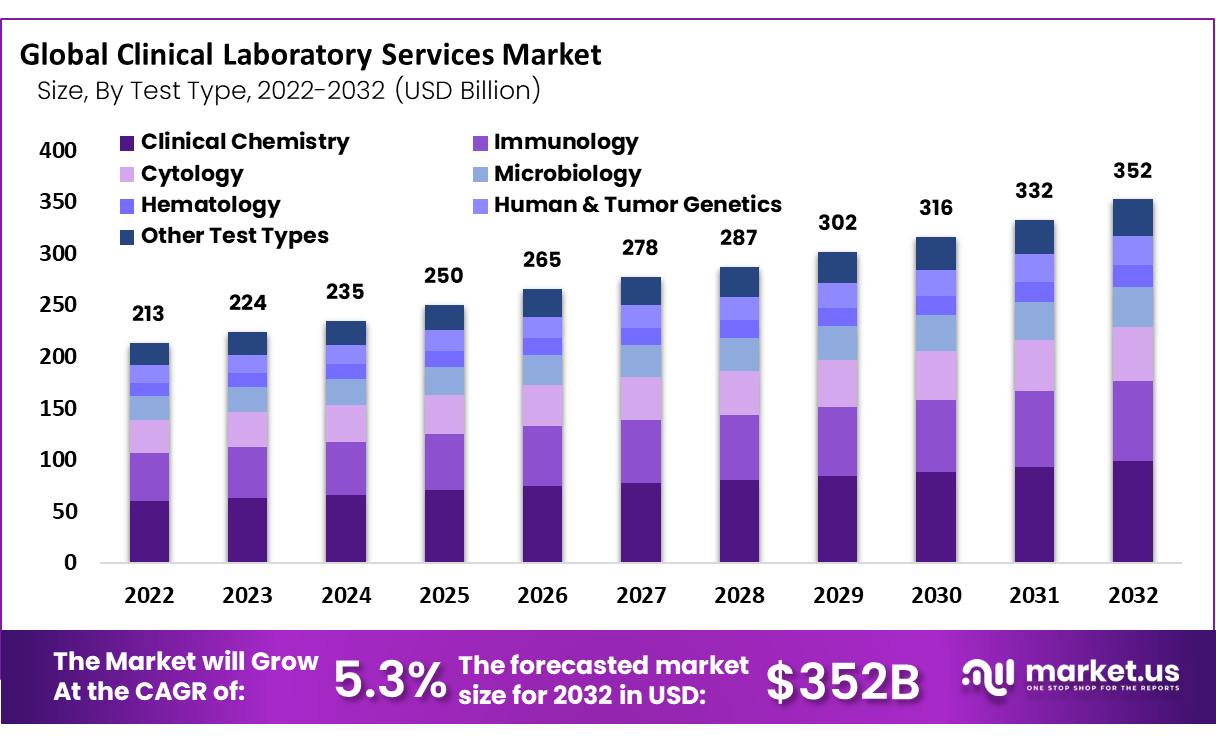
The Global Clinical Laboratory Services Market size is projected to increase at an anticipated compound annual compound growth rate of 5.3% between 2022-2032, reaching USD 213.5 Billion by 2022 from USD 352 billion currently.
Key Takeaway
- by Test Type The clinical chemistry segment accounted for the highest part of the revenue generated in the market for clinical laboratory services because of the numerous clinical chemistry tests that are involved with the analysis of pathology bodily fluids.
- by Application laboratory chemistry and bioanalytical segment accounted for the largest and most successful segment in the global market for laboratory clinical services by 2022.
- Through Services Provider Analyses The hospital-based laboratory segment is expected to grow at the fastest rate of growth in the forecasted time period.
- By 2022 North America led the market with the largest share of revenue at 44.3 percent.
- Asia Pacific will be predicted to be the fastest-growing market because of the accessibility of premium medical services and a growing awareness among the public about health benefits and positive developments in healthcare benefits.
Request For Sample Report Here: https://market.us/report/clinical-laboratory-services-market/request-sample/
Regional Snapshot
- North America:
- North America is a significant marketplace for the provision of clinical laboratory services and is driven by modern medical infrastructure, technology advancements, and a high level of healthcare expenditure.
- The United States holds a major part of the region and has a reliable network of clinical labs and an emphasis on diagnostic tests that are advanced.
- Canada can also be a factor in the growth of its market, due to the demand growing for personalized treatment and preventive medicine.
- Europe:
- Europe is home to a highly developed healthcare system, as well as an established market for laboratory services in clinical settings.
- Countries such as Germany, France, the United Kingdom, and Italy contribute significantly to the industry, aided by initiatives from the government, state-of-the-art research facilities, and an increasingly aging population.
- The region places a heavy focus on quality assurance certification, accreditation, and compliance with the regulatory requirements for laboratory service.
- Asia Pacific:
- The Asia Pacific region represents a major growth area for laboratory clinical services due to the growing number of chronic diseases as well as the development of better healthcare infrastructure and growing awareness regarding the early detection of disease.
- Countries like China, Japan, India, as well as South Korea, are driving market development, driven by an increase in spending on healthcare and the use of new laboratory techniques.
- The emerging economies of Southeast Asia, such as Indonesia, Thailand, and Vietnam have also witnessed an explosion in the demand for the use of clinical laboratory services.
- Latin America:
- Latin America is experiencing steady expansion in the field of clinical laboratory services due to the increase in investment in healthcare, increasing consciousness about the importance of preventive healthcare, and the rising prevalence of infectious diseases.
- Brazil, Mexico, and Argentina are the major regional markets that are driven by the improvement of health infrastructure, the expansion of private health care, as well as initiatives by government agencies to boost diagnostic abilities.
- Middle East and Africa:
- The Middle East and Africa region is witnessing a steady transition to advanced diagnostic tests as well as personalized medical treatment.
- countries like Saudi Arabia, UAE, and South Africa are prominent markets that are driven by the growth in healthcare spending, the increasing demand for tests that are specialized, and initiatives to improve access to healthcare as well as quality.
Drivers
- Increased Demand for Diagnostic Testing: With rising chronic and infectious disease prevalence coupled with an aging population, diagnostic testing services play a crucial role in disease detection, monitoring, treatment decision making, and decision support for clinical laboratory services.
- Technological Advancements: Recent advances in laboratory technologies like automation, molecular diagnostics, and point-of-care testing have greatly increased accuracy, efficiency, and speed for clinical laboratory services. These technologies facilitate fast test results leading to enhanced patient care.
- Focus on Personalized Medicine: As we move closer towards personalized medicine – which involves tailoring medical treatments according to each patient's genetic, environmental, and lifestyle factors – advanced diagnostic testing becomes crucial in making informed treatment decisions for personalized healthcare plans. Clinical laboratory services play a vital role in gathering critical data required for personalized therapy decisions.
- Increased Healthcare Spending: Soaring global healthcare expenditure due to expanded access and government initiatives is driving clinical laboratory service growth. Sufficient funding and reimbursement policies facilitate the adoption of laboratory tests.
- Preventive Healthcare Awareness Increases Demand: Enhanced awareness about early disease detection and preventive healthcare drives the demand for regular checkups and screening tests, leading to early diagnoses for timely interventions that lead to improved health outcomes. Clinical laboratory services play a pivotal role here.
Restraints
- Stringent Regulatory Environment: Clinical laboratory services are subject to stringent regulatory requirements, accreditation processes, and quality control standards. Compliance with these regulations can be complex and time-consuming, posing challenges for laboratory operators.
- Reimbursement Challenges: In many healthcare systems, reimbursement policies and rates for laboratory services are subject to change, affecting the financial viability of clinical laboratories. Negotiating favorable reimbursement terms and adapting to evolving payment models can be challenging.
- Cost Constraints: The high cost of advanced laboratory equipment, consumables, and skilled personnel can pose financial challenges for clinical laboratory operators. Cost containment measures and managing operational expenses are crucial to maintaining profitability.
- Shortage of Skilled Workforce: The clinical laboratory field faces a shortage of skilled laboratory professionals, including pathologists, technicians, and technologists. Addressing workforce shortages and ensuring adequate training and education programs are vital for sustained market growth.
- Data Privacy and Security Concerns: Clinical laboratory services involve handling sensitive patient data, including medical records and genetic information. Ensuring data privacy, security, and compliance with data protection regulations is essential to maintain patient trust and avoid data breaches.
- Limited Access to Advanced Technologies: In some regions, access to advanced laboratory technologies and specialized tests may be limited, particularly in rural or underserved areas. Improving infrastructure and expanding access to advanced laboratory services are important challenges to address.
Opportunities
- Rising Demand for Point-of-Care Testing: Point-of-care testing (POCT) allows for rapid diagnostics and immediate treatment decisions at the patient's bedside or in decentralized settings. Clinical laboratory services can capitalize on the growing demand for POCT by offering innovative testing solutions and expanding their service offerings.
- Growth of Genetic Testing and Precision Medicine: The field of genetic testing and precision medicine is expanding, driven by advancements in genomic technologies and an increasing understanding of the genetic basis of diseases. Clinical laboratory services can seize the opportunity by offering comprehensive genetic testing services and collaborating with healthcare providers to deliver personalized treatment plans.
- Expansion of Telemedicine and Remote Testing: The adoption of telemedicine and remote healthcare services has accelerated, enabling patients to access medical consultations and testing services remotely. Clinical laboratory services can tap into this trend by providing remote testing options, establishing partnerships with telemedicine platforms, and offering convenient sample collection methods.
- Integration of Artificial Intelligence and Data Analytics: The integration of artificial intelligence (AI) and data analytics in clinical laboratory services can unlock valuable insights from large volumes of data, leading to improved diagnostics, predictive analytics, and optimized laboratory operations. Embracing AI-driven solutions and developing advanced data analytics capabilities can create opportunities for growth and efficiency.
- Collaboration with Research Institutions and Biotech Companies: Collaboration with research institutions, biotech companies, and pharmaceutical companies presents opportunities for clinical laboratory services to participate in clinical trials, biomarker research, and drug development. Such partnerships can enhance revenue streams, foster innovation, and drive advancements in diagnostic testing.
Make an inquiry before picking up this report @ https://market.us/report/clinical-laboratory-services-market/#inquiry
Challenges
- Price Pressure and Reimbursement Uncertainty: Clinical laboratory services face ongoing pressure on pricing due to cost containment measures by payers, such as government healthcare programs and insurance companies. Maintaining profitability and navigating reimbursement uncertainties can be challenging for laboratory operators.
- Regulatory Compliance and Quality Assurance: Compliance with regulatory requirements, such as accreditation standards and quality control measures, is a critical challenge for clinical laboratory services. Ensuring adherence to changing regulations and maintaining high-quality standards across operations are essential for success.
- Evolving Technology Landscape and Investment Requirements: Rapid advancements in laboratory technologies necessitate ongoing investments in infrastructure, equipment, and skilled personnel. Staying up-to-date with emerging technologies and managing the associated investment costs pose challenges for laboratory services.
- Data Security and Privacy Concerns: Clinical laboratory services handle sensitive patient data, making data security and privacy a top concern. Safeguarding patient information, complying with data protection regulations, and implementing robust cybersecurity measures are critical challenges in the digital era.
- Workforce Shortages and Skill Gaps: The clinical laboratory field faces challenges in attracting and retaining skilled laboratory professionals. Workforce shortages and skill gaps in areas such as laboratory technicians, technologists, and pathologists can hinder operational efficiency and service delivery.
- Competitive Landscape and Consolidation: The clinical laboratory services market is highly competitive, with both large national players and local/regional laboratories vying for market share. The consolidation trend in the industry, driven by mergers and acquisitions, presents challenges for smaller or independent laboratories to compete effectively.
Key Market Players
- QIAGEN N.V.
- OPKO Health
- Siemens Medical Solutions USA, Inc.
- Quest Diagnostics
- Abbott Laboratories
- NeoGenomics Laboratories, Inc
- Fresenius Medical Care AG & Co.
- Sonic Healthcare Limited
- Associated Regional and University Pathologists, Inc.
- Laboratory Corporation of America Holdings,
- Other Key Players
Key Market Segments
Based on the Test Type
- Clinical Chemistry
- Immunology
- Cytology
- Microbiology
- Hematology
- Human & Tumor Genetics
- Endocrinology
- Other Test Types
Based on Application
- Bioanalytical & Lab Chemistry
- Cell & Gene Therapy
- Toxicology Testing
- Preclinical & Clinical Trials
- Drug Discovery & Development
Based on the Service Provider
- Standalone Laboratories
- Hospital-Based Laboratories
- Clinic-Based Laboratories
- Other Service Providers
Top Impacting Factors
- Disease Prevalence and Aging Population: The prevalence of chronic and infectious diseases, coupled with an aging population, drives the demand for clinical laboratory services. As the global population continues to age and the burden of diseases increases, the need for diagnostic testing and monitoring services grows, positively impacting the market.
- Technological Advancements: Advances in laboratory technologies, such as automation, molecular diagnostics, next-generation sequencing, and digital pathology, have revolutionized clinical laboratory services. These advancements improve the accuracy, efficiency, and speed of diagnostic testing, enabling more precise disease detection, personalized treatment approaches, and improved patient care.
- Government Initiatives and Healthcare Policies: Government initiatives aimed at improving healthcare access, quality, and affordability play a crucial role in shaping the clinical laboratory services market. Policies related to healthcare infrastructure development, reimbursement, and quality assurance impact the growth and sustainability of laboratory services.
- Focus on Preventive Healthcare and Early Disease Detection: Increasing awareness about the benefits of preventive healthcare and early disease detection drives the demand for routine check-ups, screening tests, and health monitoring. Clinical laboratory services are instrumental in providing the necessary diagnostic tests to facilitate early diagnosis and intervention, leading to better health outcomes.
- Adoption of Personalized Medicine: Personalized medicine, which involves tailoring medical treatment based on individual patient characteristics, is gaining traction. Clinical laboratory services play a vital role in providing diagnostic tests for genetic profiling, biomarker analysis, and treatment response monitoring, supporting the implementation of personalized medicine approaches.
Recent Developments
- In March 2022 Illumina Inc. introduced TruSight oncology. It is a one-time test that tests various tumor-related biomarkers and genes in order to establish the specific genetic profile of a patient.
- On October, 2021. the Cobas Pure Integrated Solutions Analyzer was developed by Roche Diagnostics, the Indian department of Roche Diagnostics, and features a higher-end system, making it suited for many laboratories and hospitals across India.
Report Scope
| Report Attribute | Details |
| The market size value in 2022 | USD 213.5 Bn |
| Revenue Forecast by 2032 | USD 352 Bn |
| Growth Rate | CAGR Of 5.3% |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, and Middle East & Africa, and Rest of the World |
| Historical Years | 2017-2022 |
| Base Year | 2022 |
| Estimated Year | 2023 |
| Short-Term Projection Year | 2028 |
| Long-Term Projected Year | 2032 |
Frequently Asked Questions
Q: What is the current size of the Clinical Laboratory Services Market?
A: The Global Clinical Laboratory Services Market size is USD 213.5 Billion in 2022.
Q: What is the projected growth rate for Clinical Laboratory Services Market?
A: The Clinical Laboratory Services Market is expected to grow at a CAGR of 5.3% from 2023 to 2032.
Q: What are some of the key players in the Clinical Laboratory Services Market?
A: Some of the key players in the Clinical Laboratory Services market include QIAGEN N.V., OPKO Health, Siemens Medical Solutions USA, Inc., Quest Diagnostics, Abbott Laboratories, NeoGenomics Laboratories, Inc, Fresenius Medical Care AG & Co., Sonic Healthcare Limited, Associated Regional and University Pathologists, Inc., Laboratory Corporation of America Holdings, Other Key Players
Contact:
Global Business Development Team – Market.us
Market.us (Powered by Prudour Pvt. Ltd.)
Send Email: [email protected]
Address: 420 Lexington Avenue, Suite 300 New York City, NY 10170, United States
Tel: +1 718 618 4351
Website: https://market.us
Content has been published via 11press. for more details please contact at [email protected]
The team behind market.us, marketresearch.biz, market.biz and more. Our purpose is to keep our customers ahead of the game with regard to the markets. They may fluctuate up or down, but we will help you to stay ahead of the curve in these market fluctuations. Our consistent growth and ability to deliver in-depth analyses and market insight has engaged genuine market players. They have faith in us to offer the data and information they require to make balanced and decisive marketing decisions.